tl;dr: sugars are examples of isomers in our bodys. IOOsomers are handled differntl
Here comes pibbur.
No,I didn't know about the two water isomers. But I know a bit or two about biologically important isomers.
Let's consider sugar. (The following is a simplified explanation, leaving out a lot of details.)
There are large sugar molecules like starch and cellulose. These are long chains of simpler building block called
monosaccarides. The most important monosaccaride is
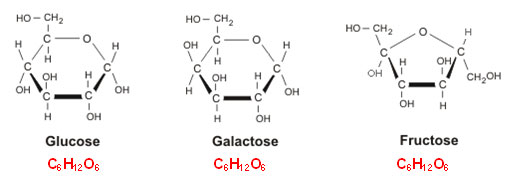
glucose, which has the general formula C6H12O6, 6 carbon atoms, 12 hydrogens and 6 oxygen atoms. Glucose is an example of a
hexose (from hexa, 6), a sugar with 6 carbon atoms. There are however other hexoses , the most important of these are
fructose and
galactose. These have the same general formula, but hey differ from each other in how the atoms are arranged.
It's easy to see the difference between fructose and the other two, since fructose has a 5-ring, the other to are 6-ring structures. The difference between glucosis and galactosis is more subtle, but if we look at the leftmost carbon atom we see that the OH-group is directed upwards for galactose and downwards for glucose, just a small small variation but as we shall see, biochemically very important. These 3 molecules are isomers, more specifically
structural isomers.
The reason why these differences matter is how enzymes work. Enzymes are very specific when it comes to the substances they can work on (and what they can do with them). Breaking down glucose in our cells is a chain reaction with around 20 separate steps, all controlled by it's own enzyme. The enzyme
glucokinase takes care of the first step in this process. This enzyme can only handle glucose, not fructose or galactose which therefore are handled differently, but both fructose and galactose can be transformed to glucose.
We see another type of isomerism if we look closer at the glucose molecule. Turns out there are two forms of glucose,
D-glucose and
L-Glucose. These are mirror images of each other, and like other mirror images, you can't get one form from the nother just by rotating it. The D-form and L-form are
stereoisomers. Only the D-form occurs naturally, because - surprise, surprise - our enzymes can only handle the D-form. The L-form has to be made in a lab.
There are other types of monosakkarides. A
pentose is a sugar molecule with 5 carbon atoms (penta=5). One of the important pentoses is
deoxyribose, which together with phosphoric acid construct the backbone of our DNA molecules (DNA stands for
deoxyribo-nucleic acid). Pentoses and hexoses are not isomers since the numbers of atoms differ.
pibbur who observes that some of his typos can be considered isomers, since all the required letters (and no others) are there, it's just the sequence that differ. Not all of them, though. *sighs* And this is a very long rant.

PS. Alrik: I see that some of my responses to several of your posts, have been a bit patronizing and arrogant. I'm sorry for that. That was not my intention. Friends again?DS.